Phosphocreatine system[edit]
Creatine, which is synthesized in the
liver and
kidneys, is transported through the blood and taken up by tissues with high energy demands, such as the brain and skeletal muscle, through an active transport system. The concentration of
ATP in skeletal muscle is usually 2-5 mM, which would result in a muscle contraction of only a few seconds.
[13] Fortunately, during times of increased energy demands, the phosphagen (or ATP/PCr) system rapidly resynthesizes ATP from
ADP with the use of
phosphocreatine (PCr) through a reversible reaction with the enzyme
creatine kinase (CK). In skeletal muscle, PCr concentrations may reach 20-35 mM or more. Additionally, in most muscles, the ATP regeneration capacity of CK is very high and is therefore not a limiting factor. Although the cellular concentrations of ATP are small, changes are difficult to detect because ATP is continuously and efficiently replenished from the large pools of PCr and CK.
[13] Creatine has the ability to increase muscle stores of PCr, potentially increasing the muscle’s ability to resynthesize ATP from ADP to meet increased energy demands.
Crossbridge cycling[edit]
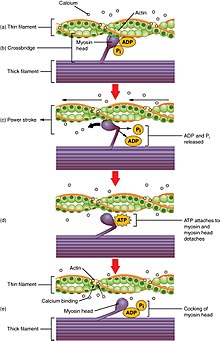
Crossbridge cycling
Crossbridge cycling is a sequence of molecular events that underlies the sliding filament theory. A crossbridge is a myosin projection, consisting of two myosin heads, that extends from the thick filaments.
[1] Each myosin head has two binding sites: one for
ATP and another for actin. The binding of ATP to a myosin head detaches myosin from
actin, thereby allowing myosin to bind to another actin molecule. Once attached, the ATP is hydrolyzed by myosin, which uses the released energy to move into the "cocked position" whereby it binds weakly to a part of the actin binding site. The remainder of the actin binding site is blocked by tropomyosin.
[24] With the ATP hydrolyzed, the cocked myosin head now contains ADP + Pi. Two Ca2+
ions bind to
troponin C on the actin filaments. The troponin-Ca2+
complex causes
tropomyosin to slide over and unblock the remainder of the actin binding site. Unblocking the rest of the actin binding sites allows the two myosin heads to close and myosin to bind strongly to actin.
[24] The myosin head then releases the inorganic phosphate and initiates a
power stroke, which generates a force of 2 pN. The power stroke moves the actin filament inwards, thereby shortening the
sarcomere. Myosin then releases ADP but still remains tightly bound to actin. At the end of the power stroke, ADP is released from the myosin head, leaving myosin attached to actin in a rigor state until another ATP binds to myosin. A lack of ATP would result in the rigor state characteristic of
rigor mortis. Once another ATP binds to myosin, the myosin head will again detach from actin and another crossbridges cycle occurs.
Crossbridge cycling is able to continue as long as there are sufficient amounts of ATP and Ca2+
in the cytoplasm.
[24] Termination of crossbridge cycling can occur when Ca2+
is
actively pumped back into the sarcoplasmic reticulum. When Ca2+
is no longer present on the thin filament, the tropomyosin changes conformation back to its previous state so as to block the binding sites again. The myosin ceases binding to the thin filament, and the muscle relaxes. The Ca2+
ions leave the troponin molecule in order to maintain the Ca2+
ion concentration in the sarcoplasm. The active pumping of Ca2+
ions into the sarcoplasmic reticulum creates a deficiency in the fluid around the myofibrils. This causes the removal of Ca2+
ions from the troponin. Thus, the tropomyosin-troponin complex again covers the binding sites on the actin filaments and contraction ceases.
That's why Calcium ion concentration is important and eating chelating agents such as casein, coca cola e.t.c. will reduce the Ca2+ content but I don't know if it will reduce the Ca2+ plasma content, I'm not a biologist or a medic.



